Buy Tesamorelin Peptide at Wholesale Peptide
Molecular Structure of Tesamorelin
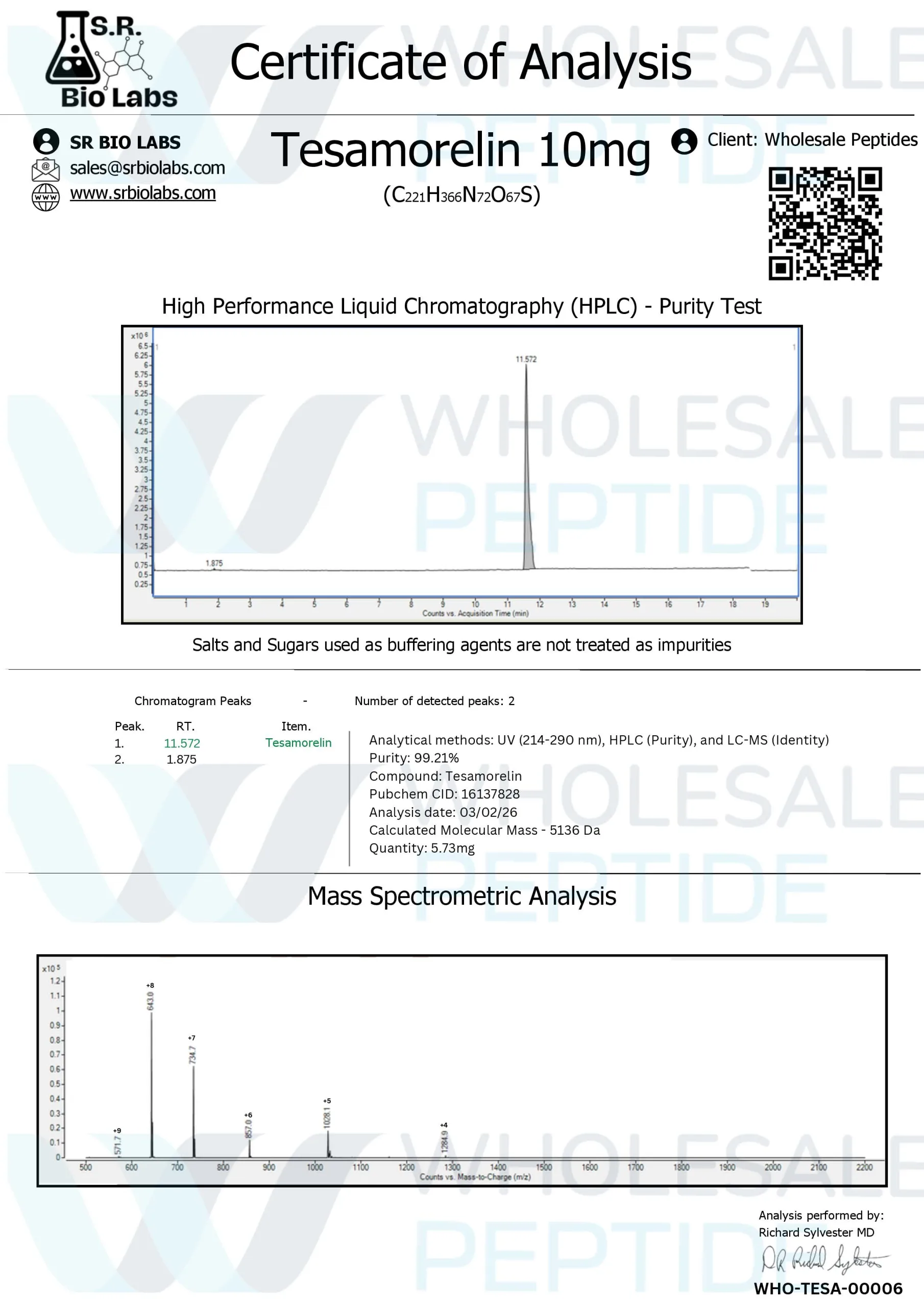
Amino Sequence: 44-amino acid synthetic growth hormone releasing factor analogue with trans-3-hexenoic acid modification at N-terminal
Molecular formula: C₂₂₁H₃₆₆N₇₂O₆₇S
Molecular Weight: 5136 g/mol
Chemical name: (3E)-hex-3-enoylsomatoliberin
CAS #: 218949-48-5
Tesamorelin is a synthetic polypeptide designed as a hormone analogue of human growth hormone releasing factor (GHRH). The peptide has enhanced stability compared to native GHRH due to N- and C-terminus modifications, making it more resistant to enzymatic degradation. The trans-3-hexenoic acid modification increases both the stability and potency of tesamorelin compared to endogenous GHRH, with a half-life of approximately 26-38 minutes when administered subcutaneously.
Mechanism of Action
Tesamorelin is a growth hormone releasing factor analogue that acts specifically on GHRH receptors located on somatotroph cells within the anterior pituitary gland. This binding activates G-protein-coupled receptor pathways, leading to increased synthesis and pulsatile release of endogenous human growth hormone while maintaining physiological feedback patterns.
The released growth hormone then stimulates hepatocytes to increase production of insulin like growth factor-1 (IGF-1) and IGF-binding protein 3 (IGFBP-3), both contributing to anabolic effects and tissue growth. The lipolytic action of growth hormone, enhanced by tesamorelin, activates hormone-sensitive lipase and promotes fatty acid oxidation, preferentially targeting visceral fat reduction while generally preserving subcutaneous fat and lean muscle mass.
This selective mechanism makes tesamorelin great for reducing excess abdominal fat and improving body composition, especially in conditions involving abnormal fat distribution such as hiv associated lipodystrophy.
Research Studies
Extensive research has been done on tesamorelin. Phase III placebo controlled trial studies with 806 hiv infected patients with lipodystrophy showed significant visceral fat reduction, with subjects losing an average of 15.4% of excess fat over several months of treatment.
Here are the key findings:* HIV-Associated Lipodystrophy Trials: Studies in hiv patients on antiretroviral therapy showed significant reduction in excess abdominal fat and metabolic improvements including triglyceride levels and lipid metabolism
- Hepatic Fat Studies: 12-month studies in hiv positive subjects showed decreased liver fat and metabolic parameters
- Muscle Composition Research: CT scan analysis showed increased muscle density and muscle area, decreased muscle fat in paraspinal muscles and psoas major
- Insulin Sensitivity Trials: 12-week studies showed improved insulin resistance and glucose intolerance in infected patients
- Safety Extension Data: Long term monitoring showed no drug induced liver injury or internal organ toxicity
- Cognitive Function Research: Emerging studies in j frailty aging populations suggest benefits for mild cognitive impairment associated with metabolic dysfunction
The primary outcome across multiple studies is tesamorelin reduces fat while maintaining safety profile vs placebo group.
Why Buy from Wholesale Peptide
Wholesale Peptide offers high purity tesamorelin peptide for research use. Our wholesale pricing is designed for bulk orders for extended research and consistent supply chain for ongoing studies.All tesamorelin products sold by Wholesale Peptide are research chemicals only. This designation allows the use of this synthetic growth hormone releasing peptide for in-vitro laboratory testing and experimentation only. Human or veterinary use is strictly forbidden. This product is not a drug, food or cosmetic and may not be misbranded, mislabeled or misused as such.
Research applications may include studying the peptide’s effects on fat content, body composition, programmed cell death pathways and metabolic processes but must remain within approved research parameters.
Shipping Policy of Wholesale Peptide
Wholesale Peptide ships same day for orders placed before 1pm EST Monday through Friday. Orders placed after 1pm EST or on weekends will be shipped the next business day. Temperature controlled shipping ensures peptide stability during transport, with tracking information provided for all shipments to maintain research continuity.
Satisfaction Guarantee
Wholesale Peptide offers 30 day satisfaction guarantee on all tesamorelin products. Simply return any unopened products to us for full refund of the purchase price of unused materials. Our quality assurance commitment supports research applications with dedicated customer service for any product concerns.
Third Party Testing of Every Batch
Every batch of tesamorelin is tested by third party using HPLC to ensure product purity and accuracy. Certificate of analysis is provided with each batch verifying molecular weight and amino acid sequence. Quality control measures ensure research grade standards for studying tesamorelin’s effect on growth hormone pathways and metabolic processes.
References and Citations
- Falutz J, Allas S, Kotler D, et al. “A placebo-controlled, dose-ranging study of a growth hormone releasing factor in HIV-infected patients with abdominal fat accumulation.” AIDS. 2005;19(12):1279-1287.
- Falutz J, Allas S, Blot K, et al. “Metabolic effects of a growth hormone-releasing factor in patients with HIV.” New England Journal of Medicine. 2007;357(23):2359-2370.
- Falutz J, Mamputu JC, Potvin D, et al. “Long-term safety and effects of tesamorelin, a growth hormone-releasing factor analogue, in HIV patients with abdominal fat accumulation.” AIDS. 2008;22(14):1719-1728.* Stanley TL, Falutz J, Marsolais C, et al. “Visceral fat reduction is associated with improved metabolic profile in HIV-infected patients treated with tesamorelin.” Clinical Infectious Diseases. 2012;54(12):1642-1651.
- Makimura H, Feldpausch MN, Rope AM, et al. “Metabolic effects of a growth hormone-releasing factor in obese subjects with reduced growth hormone secretion: a randomized controlled trial.” Journal of Clinical Endocrinology & Metabolism. 2012;97(12):4769-4779.
- Clemmons DR, Miller S, Mamputu JC. “Safety and metabolic effects of tesamorelin, a growth hormone-releasing factor analogue, in patients with type 2 diabetes: A randomized, placebo-controlled trial.” PLoS One. 2017;12(6):e0179538.
- Fourman LT, Czerwonka N, Feldpausch MN, et al. “Visceral fat reduction with tesamorelin is associated with improved liver enzymes in HIV.” AIDS. 2017;31(16):2253-2259.
- U.S. Food and Drug Administration. “Egrifta (tesamorelin) injection approval letter.” 2010.
- National Institute of Diabetes and Digestive and Kidney Diseases. “Clinical and research information on tesamorelin and lipodystrophy.” Accessed 2024.
- Spooner LM, Olin JL. “Tesamorelin: a growth hormone-releasing factor analogue for HIV-associated lipodystrophy.” Annals of Pharmacotherapy. 2012;46(2):240-247.
| Weight | .03125 lbs |
|---|