Buy Tesamorelin at Wholesale Peptide
Research into growth hormone releasing mechanisms has led to many advances in peptide pharmacology and metabolic regulation. Tesamorelin, a synthetic hormone analogue of human growth hormone releasing factor, is a research compound that has gained a lot of scientific interest for its unique molecular properties and research applications in lab settings.
As researchers buy tesamorelin for their research, understanding its molecular characteristics, mechanism of action and handling protocols becomes crucial for conducting scientific studies. This overview covers the structural features and research applications of this growth hormone releasing hormone analogue.
Molecular Structure of Tesamorelin
Tesamorelin has a complex peptide structure that distinguishes it from natural growth hormone releasing factor. The compound’s amino acid sequence shows the design of this hormone releasing factor analogue:
Amino Sequence: Tyr-Ala-Asp-Ala-Ile-Phe-Thr-Asn-Ser-Tyr-Arg-Lys-Val-Leu-Gly-Gln-Leu-Ser-Ala-Arg-Lys-Leu-Leu-Gln-Asp-Ile-Met-Ser-Arg-Gln-Gln-Gly-Glu-Ser-Asn-Gln-Glu-Arg-Gly-Ala-Arg-Ala-Arg-Leu-NH2
Molecular Formula: C221H366N72O67S1
Molecular Weight: 5135.77 g/mol
PubChem CID: 16132269
CAS #: 218949-48-5
The modifications in tesamorelin include an N-terminal trans-3-hexenoyl group which research shows enhances stability compared to native growth hormone releasing hormone. This modification reduces enzymatic degradation making it very useful for lab research on prolonged growth hormone releasing mechanisms.
Mechanism of Action
Research has shown that tesamorelin acts as a synthetic analogue of human hypothalamic growth hormone releasing factor. The compound binds to GHRH receptors on pituitary gland somatotrophs and triggers a cascade of physiological responses in experimental models.
Lab research has shown that tesamorelin stimulates the synthesis and pulsatile release of natural growth hormone production by interacting with the anterior pituitary gland. This leads to increase in insulin like growth factor-1 (IGF-1) and its binding protein IGFBP-3 in research subjects.The mechanism involves lipolysis which research shows breaks down stored triglycerides in visceral adipose tissue. Studies have focused on its effects on excess abdominal fat and visceral fat reduction in experimental models. Research shows this tesamorelin therapy approach supports natural growth hormone production pathways while keeping the body’s endogenous hormone axis intact.
Research into lipid metabolism has shown the compound affects body composition through its action on growth hormone releasing pathway. Research suggests these metabolic effects may impact muscle density, liver fat content and overall fat distribution in lab studies.
Research Studies
Scientific research into tesamorelin has focused on its fda approved application for excess fat accumulation, particularly in hiv associated lipodystrophy models. Multiple placebo controlled trial designs have looked at the compound’s effects on visceral adipose tissue over 12-26 weeks.
Research findings show statistically significant reduction in visceral fat when comparing treatment group to placebo group. These measurements are typically obtained through computed tomography or MRI imaging and provide quantitative data on internal organs fat distribution and paraspinal muscles composition.
| Study Parameter | Treatment Group | Control Group | Clinical Significance |
|---|---|---|---|
| Visceral fat reduction | Significant decrease | No change | Clinically meaningful differences |
| IGF-1 levels | Elevated | Baseline | Consistent with mechanism |
| Study duration | 12-26 weeks | Same period | Adequate assessment time |
Pooled analysis of multiple studies has shown correlation between treatment and increased IGF-1 levels, supporting the compound’s mechanism of action. However, research has also shown glucose intolerance and fasting glucose levels in some experimental models.
Safety evaluation studies have looked at injection site reactions, joint pain and peripheral edema in research subjects. The safety extension data from longer-term studies provides valuable information for researchers designing longer study protocols.
Studies on muscle area measurements, including rectus abdominis and psoas major regions, have shown significant improvement in muscle fat ratios and overall body composition parameters. These findings contribute to understanding the compound’s broader metabolic effects beyond visceral fat reduction.
Research in older adults population has looked at potential application in hormone replacement therapy context, though these studies are in early phase. The national institute has funded various research projects on the compound’s effects on energy levels and overall well being in controlled lab settings.
Storage and Safety
Proper handling protocols are important when working with tesamorelin in research settings. The compound is usually supplied as a lyophilized powder and requires specific storage conditions to maintain stability and integrity for lab use.
Storage requirements include refrigeration (no freezing), protection from light and tight sealed containers. The reconstituted solution requires similar storage conditions and should be used within timeframe specified by the manufacturer.
Lab safety protocols recommend all handling procedures follow established guidelines for research compounds. Disposal of unused materials should be done through waste management programs to prevent environmental contamination and comply with institutional safety requirements.
Research facilities should keep record of storage conditions and handling procedures as part of their quality assurance protocols. Temperature monitoring and environmental controls ensure compound stability throughout the research period.
Why Buy from Wholesale Peptide
Wholesale Peptide has several advantages for researchers looking to buy tesamorelin for their lab studies. Their operational framework addresses key concerns that research scientists face when sourcing high quality peptide compounds.
Shipping Policy of Wholesale Peptide
Wholesale Peptide ships same day for orders placed before 1pm EST Monday through Friday. Orders placed after 1pm EST or weekends will be shipped the next business day. This fast turnaround supports time sensitive research protocols and keeps project timelines.
Satisfaction Guarantee
Wholesale Peptide offers 30 day satisfaction guarantee on all products purchased from us. Simply return any unopened products to us for full refund of the purchase price of the unused products. This policy gives researchers confidence in their procurement decisions and reduces financial risk associated with compound sourcing.
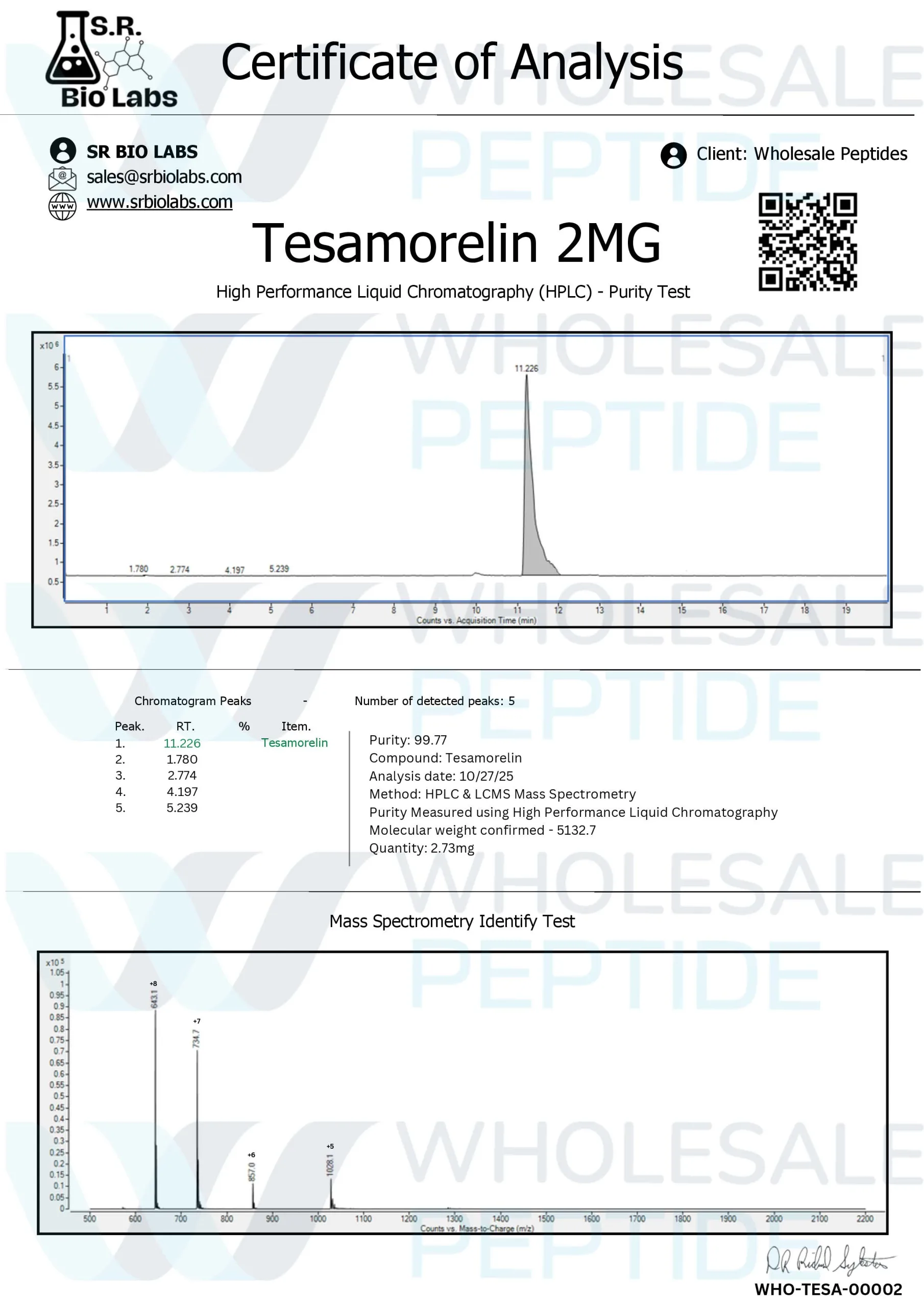
Third Party Testing of Every Batch
Every batch of products sold by Wholesale Peptide is tested by third party using HPLC to ensure product purity and accuracy. This quality control measure is important for research applications where compound purity directly impacts experimental validity and reproducibility.
The HPLC testing verifies both identity and purity specifications, provides researchers with documentation for regulatory compliance and scientific rigor. This testing supports non clinical pharmacology studies that require compound characterization.
Quality control goes beyond basic purity testing to include verification of molecular integrity and stability parameters. These comprehensive testing protocols ensure researchers receive compounds suitable for their specific experimental requirements.
Products from Wholesale Peptide are for Research Use Only
All products from Wholesale Peptide are research chemical only. This means use of this chemical is strictly for in-vitro laboratory testing and experimentation. Human or veterinary use is strictly forbidden. This product is not a substance, food or cosmetic and may not be misbranded, mislabeled or misused as such.
The research use only classification complies with federal regulations governing the distribution and use of investigational compounds. This classification protects both suppliers and researchers by clearly defining appropriate applications and preventing misuse of research materials.
Researchers buying tesamorelin must show legitimate research purposes and keep proper documentation of their studies. This requirement supports the ongoing research while ensuring regulatory compliance throughout the research process.
The compound is classified under research use only guidelines means any use must be within approved laboratory settings under supervision. This framework supports the integrity of research while keeping clear boundaries of use.
Lab protocols should include proper documentation of research objectives, methodology and safety procedures when working with research grade tesamorelin. These requirements ensure research is conducted according to scientific standards and regulatory guidelines.
Understanding the difference between research compounds and other substance classification is important to comply with drug class regulations and avoid legal issues. Researchers should familiarize themselves with all applicable guidelines before starting studies with synthetic hgh analogues or related growth hormone releasing compounds.
This comprehensive approach to compound classification and distribution supports the advancement of scientific knowledge while keeping research integrity and regulatory compliance in check.
References
- Falutz J, Allas S, Kotler D, et al. A placebo-controlled, dose-ranging study of a growth hormone releasing factor in HIV-infected patients with abdominal fat accumulation. AIDS. 2005;19(12):1279-1287.
- Falutz J, Allas S, Blot K, et al. Metabolic effects of a growth hormone-releasing factor in patients with HIV. N Engl J Med. 2007;357(23):2359-2370.
- Falutz J, Mamputu JC, Potvin D, et al. Long-term safety and effects of tesamorelin, a growth hormone-releasing factor analogue, in HIV patients with abdominal fat accumulation. AIDS. 2008;22(14):1719-1728.4. Stanley TL, Feldpausch MN, Oh J, et al. Tesamorelin and visceral fat and liver fat in HIV-infected patients with abdominal fat accumulation: a randomized clinical trial. JAMA. 2014;312(4):380-389.
- Fourman LT, Czerwonka N, Feldpausch MN, et al. Visceral fat reduction with tesamorelin is associated with improved liver enzymes in HIV. AIDS. 2017;31(16):2253-2259.
- Clemmons DR, Miller S, Mamputu JC. Tesamorelin, a growth hormone-releasing factor analogue, in patients with type 2 diabetes: A randomized, placebo-controlled trial. PLoS One. 2017;12(5):e0179538.
- Grunfeld C, Dritselis A, Kirkpatrick P. Tesamorelin. Nat Rev Drug Discov. 2011;10(2):95-96.
- Chalasani N, Bonkovsky HL, Fontana R, et al. Features and outcomes of 899 patients with drug-induced liver injury: The DILIN Prospective Study. Gastroenterology. 2015;148(7):1340-1352.e7.
- The U.S. Food and Drug Administration. Egrifta (tesamorelin) prescribing information. 2010.
- Stanley TL, Falutz J, Marsolais C, et al. Visceral fat reduction is associated with improved metabolic profile in HIV-infected patients receiving tesamorelin. Clin Infect Dis. 2012;54(12):1642-1651.
| Weight | .001 lbs |
|---|